Bipolar Electrodialyzer (EDBM)
Two chambers(Doublet) Electrodialyzer BPED
In a Bipolar Electrodialyzer (EDBM), a triplet (also called a "cell" or "repetitive unit") is the fundamental repeating unit of a 2-compartment (chambers) membrane stack.
In Bipolar Membrane Electrodialysis (BMED), a doublet is a simplified, 2-compartment (two chambers)repeating unit. Unlike the 3-compartment triplet, which separates the salt feed, acid product, and base product into three distinct channels, the doublet merges the salt with either the acid or the base.
This configuration is specifically used when you only need to recover one high-purity product (either the acid or the base) or when the salt itself is an organic acid/base salt.
The Two Types of Doublets
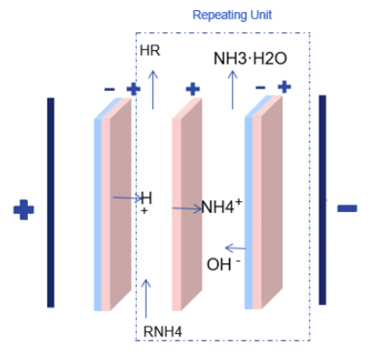
1,BPM-CEM Doublet (Base Recovery)
Composition: Bipolar Membrane (BPM) + Cation Exchange Membrane (CEM).
The Process:
The salt is fed into a compartment where it is converted into a mixture of acid and salt.
Cations Na+, K+ migrate through the CEM into a separate compartment.
Hydroxide ions OH- from the BPM enter this second compartment, forming pure base (e.g., NaOH).
Primary Use: Converting organic salts (like sodium acetate) into free acid and recovering sodium hydroxide.
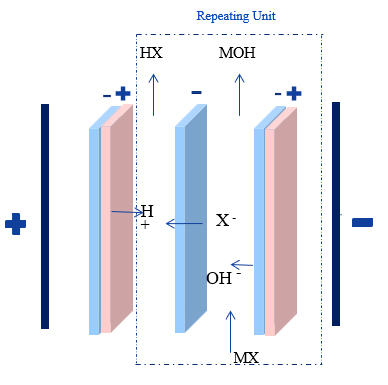
BPM-AEM Doublet (Acid Recovery)
Composition: Bipolar Membrane (BPM) + Anion Exchange Membrane (AEM).
The Process:
The salt is fed into a compartment where it becomes a mixture of base and salt.
Anions Cl-, SO42- migrate through the AEM into a separate compartment.
Protons H+ from the BPM enter this second compartment, forming pure acid (e.g., HCl).
Primary Use: Production of fine chemicals or recovering acids from organic base salts.
Working Principle
Water Dissociation: When a direct current is applied, water molecules at the interface of the BPM layers split into protons H+and hydroxide ions OH-.
Ion Migration: The applied electric field forces cations from the salt channel through the CEM into the base channel, and anions through the AEM into the acid channel.
Chemical Production: The migrated salt ions combine with the generated H+ andOH- to produce concentrated acid and base solutions.
Why Choose a Doublet(two chamber BPED)?
Doublets are the preferred choice in specific industrial scenarios:
pH Adjustment (Food & Beverage): Used in the wine industry to adjust acidity. Since the "salt" (the wine) doesn't need to be separated into two pure chemical streams, a 2-compartment system is more efficient.
Organic Acid Recovery: In fermentation (e.g., lactic acid production), the goal is often to convert the lactate salt into lactic acid. The doublet is more energy-efficient for this "electro-acidification" process.
Organic Alkali Recovery.
Simplified Plumbing: With only two liquid loops (instead of three), the pumps, tanks, and piping are significantly cheaper and easier to maintain.
Comparison: Triplet vs. Doublet
| Feature | Triplet (3-Compartment) | Doublet (2-Compartment) |
| Membranes | BPM + AEM + CEM | BPM + (AEM or CEM) |
| Output | Pure Acid and Pure Base | Acid/Salt mixture or Base/Salt mixture |
| Complexity | Higher (3 pumps/loops) | Lower (2 pumps/loops) |
| Best For | High-value chemical recovery | Simple pH adjustment |
Applications
Wastewater Treatment: Desalination and recovery of valuable resources.
CO2 Capture: Converting captured CO2 into usable products.
Ammonia Nitrogen Recovery
Our BPED Type
BPED cells: one cell=1pcs AEM + 1pcs BPM or 1pcs BPM + 1pcs CEM
60cells: generally for pilot testing
120 cells: for Industry scale
240 cells: for Industry scale
Our membrane size: 80*40cm, 110*55cm
Transportation
We will choose the suitable transportation mode and deliver the goods to you on time and safety